Cyclohexane is a cyclic (closed loop) saturated hydrocarbon. The C-C bond length of cyclohexane is too long for the molecule to fit in to one plane. To minimize bond strain, the most common conformations of cyclohexane have the C1 and C4 carbons either above or below the other carbons in the plane of the created by the other 4 carbons in the cyclohexane molecule. The most stable conformation of cyclohexane places the C1 carbon above the plane and the C4 carbon below the plane. This is called chair conformation (see fig. 2). Another prominent structure for cyclohexane places the C1 and C4 carbons both above the plane of the other carbons. This is called boat formation (see fig. 3). The boat formation is not preferred because the hydrogens on accompanying carbon atoms eclipse one another and two hydrogens attached to the C1 and C4 also interact. This is alleviated by putting the cyclohexane in chair position so that the hydrogens are not anti in position and the C1 and C4 hydrogens do not interact.
Cyclohexane Structure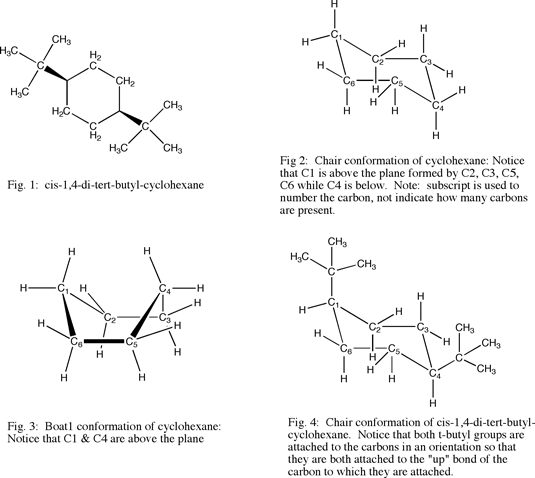
Cis-1,4-di-tert-butyl-cyclohexane has tert-butyl groups attached to the one and four carbons. Both of the t-butyl groups in the compound are attached so that they both are relatively pointed in the same direction (see fig. 4). In this exercise, the structure for cis-1,4-di-tert-butyl-cyclohexane (fig. 1) was pasted into Chem 3D. Chem 3D produced a chair conformation of the compound (see fig. 5, 7). Looking at the 3D structure reveals that there is a significant electron- electron repulsion between nonbonded atoms. Energy minimization produced a structure that strained traditional bond angles, but resulted in greater separation between molecules (see fig. 6, 8 ). This change in conformation was significant in terms of the amount of reduced energy according to Chem 3D. The steric energy of the original conformer before minimization was 520.8 kcal/mol. The steric energy after minimization was significantly reduced. Chem 3D analyzed it to be 25.2 kcal/mol. This represents a more than 20 fold decrease in strain.
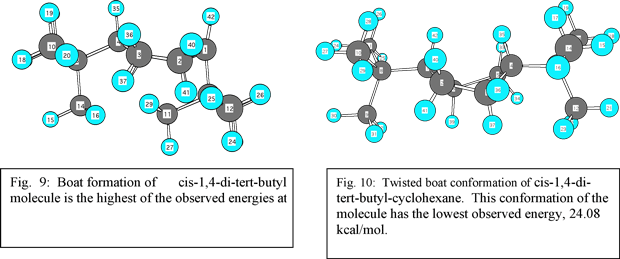
While the boat conformation of cyclohexane is the most stable form of cyclohexane, the two t-butyl groups may interact with the molecule so that there is a more stable boat formation. When cis-1,4-di-tert-butyl-cyclohexane is forced to take a boat conformation, in Chem 3D, the steric energy is extremely high, 575 kcal/mol (see fig. 9). When energy is minimized using Chem 3D an altogether different picture emerges. A twisted boat conformation appears (fig 10). This was the lowest energy conformer that could be created, 24.01 kcal/mol.
3-D Images of Chair Conformation of cis-1,4-di-tert-butyl-cyclohexane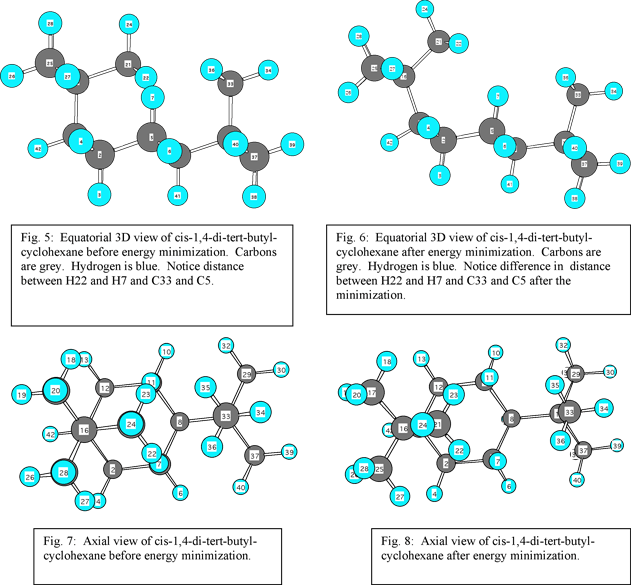 3D Images of Boat Conformation of cis-1,4-di-tert-butyl-cyclohexane
3D Images of Boat Conformation of cis-1,4-di-tert-butyl-cyclohexane